Product Brochures
Resources
25
Product Brochures
On-site animal feed analysis solutions
Application Note
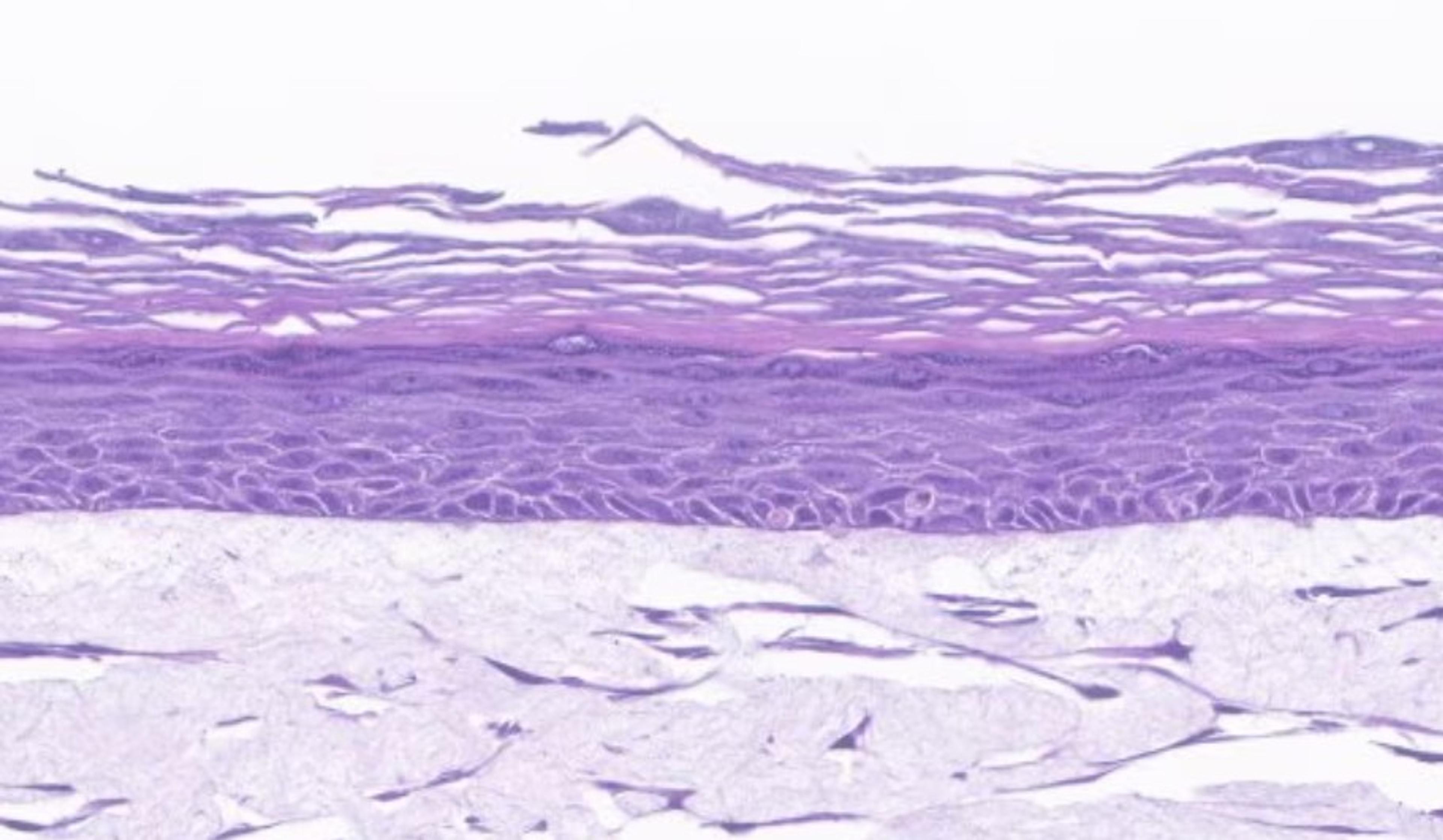
Organotypic air-liquid interface 3D cultures as in vitro models
Scientific Poster
Periodic table of antibodies
Application Note
Ensuring ADC bioanalysis accuracy
Application Note
Children's products testing services
Application Note
Environment testing services
Application Note
Material testing services
Application Note
Building product testing services
Application Note
Household and apparel product testing services
Application Note
Stationery and office supplies testing services
Application Note
7 ways to study protein phosphorylation
Product Brochures
MSDRx® Infinity data reduction software user guide
Product Brochures
MSDRx® Infinity data reduction software FAQs
Product Brochures
MSDRx® Infinity data reduction software
Application Note
Guide to western blotting
Application Note
Real-time reaction analysis
Application eBook
Cutting-edge glycan analysis to transform biopharma and diagnostics
Application Note
Fine chemical products testing services
Application Note