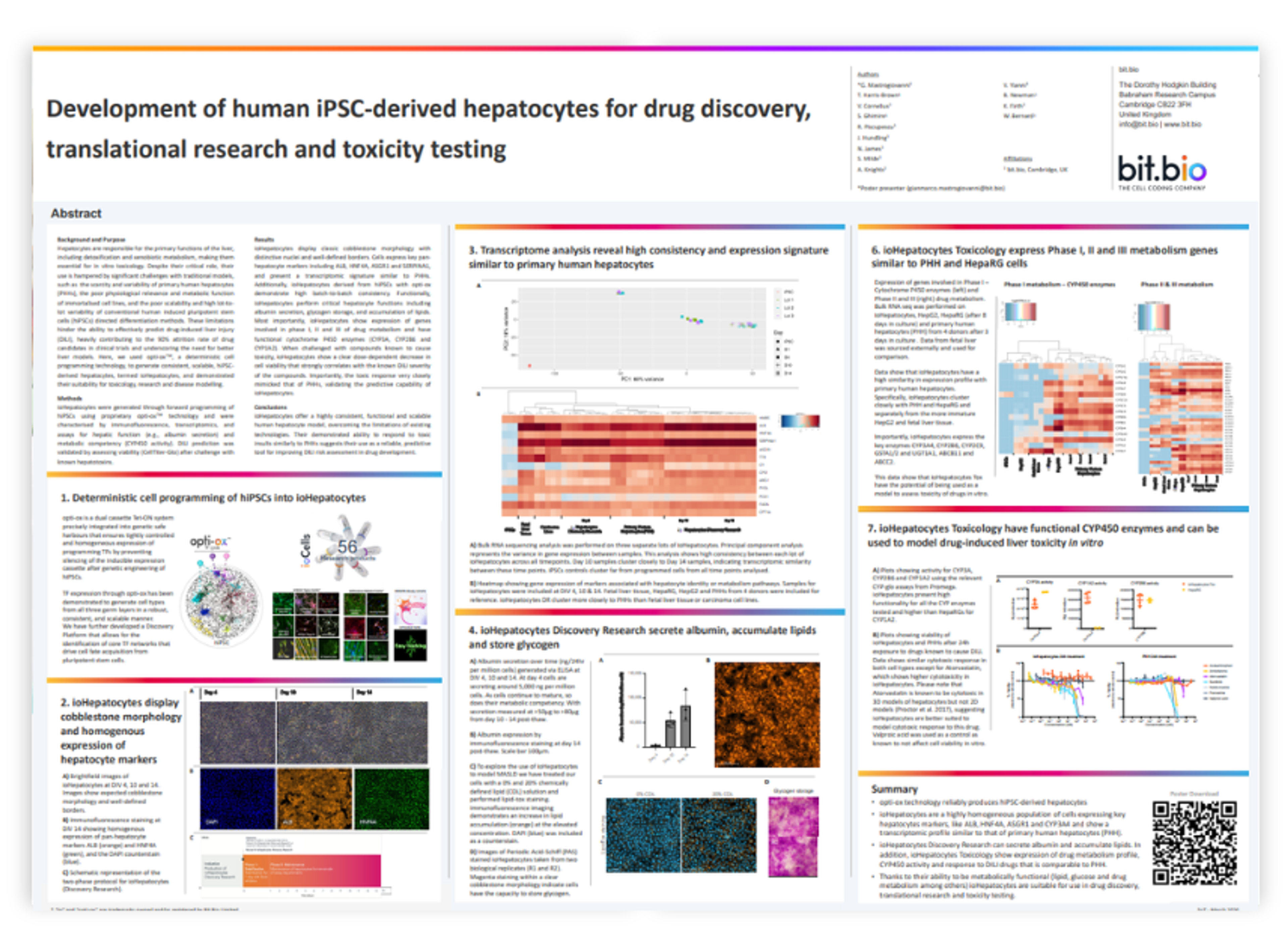
Resources
25
Selected Filters:
White Papers
The next phase of innovation in ADCs
Application Note
Critical DMPK and toxicology insights for oncology teams
Product Brochures
Percepta flyer
Application Note
Deepening digitalization in drug discovery at PTC therapeutics
Application Note
Improving pKa prediction accuracy for PROTACs
Application Note
MassivEV EV Purification Column PS instruction manual
Application Note
A new way to study cytokine release syndrome
Product Brochures
Compare humanized mouse models
Application Note
In vivo safety and efficacy platform for immunotherapies
White Papers
Antibody drug conjugates
White Papers
CNS drug development
White Papers
Human AME (mass balance) studies
White Papers
Pharmacokinetic studies in patients
White Papers
Plasma protein binding
White Papers